More life for living
MONALEESA-7: KISQALI + NSAI + goserelin in 1L premenopausal patients
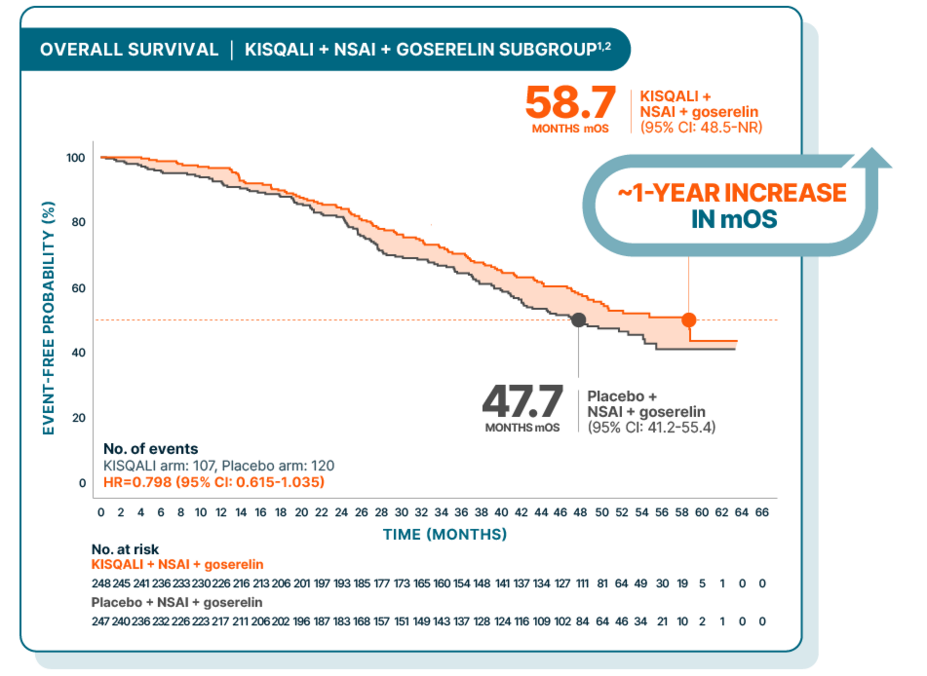
OVERALL SURVIVAL
Nearly 5 years median overall survival for 1L premenopausal patients—which means MORE life for living
At a median follow-up of 54 months
Hazard ratio is based on unstratified Cox model.2
Results from the 54-month analysis were not prespecified and were observational in nature; as such, there was no prespecified statistical procedure controlling for type 1 error
At a median follow-up of 35 months:
mOS was NR (95% CI: NR-NR) with KISQALI + NSAI + goserelin vs 40.7 months (95% CI: 37.4-NR) with placebo + NSAI + goserelin; HR=0.699 (95% CI: 0.501-0.976)3,4
Statistical significance was established for OS in the 1L ITT population; P=0.00973 (HR=0.71 [95% CI: 0.54-0.95])3
In the subgroup of patients who received tamoxifen, an increased risk for QT prolongation was observed. KISQALI is not indicated for concomitant use with tamoxifen4
1L, first line; HR, hazard ratio; ITT, intent to treat; mOS, median overall survival; NR, not reached; NSAI, nonsteroidal aromatase inhibitor; OS, overall survival.
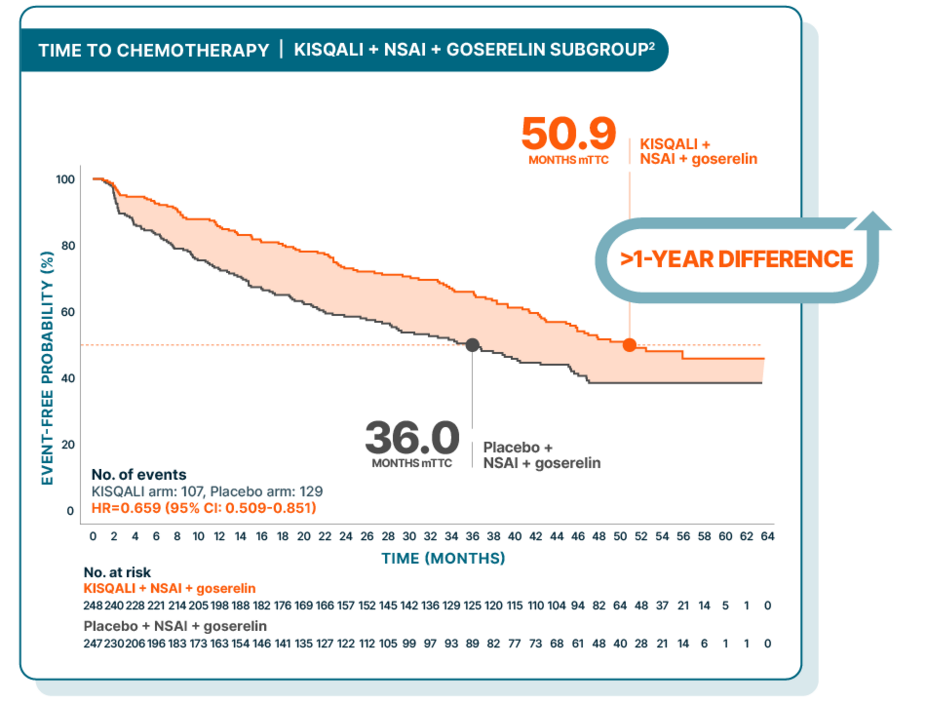
TIME TO CHEMOTHERAPY
MONALEESA-7: Median time to chemotherapy delayed over 4 years for 1L premenopausal patients
At a median follow-up of 54 months
At a median follow-up of 54 months, mTTC was 50.9 months with KISQALI + NSAI + goserelin vs 36.0 months with placebo + NSAI + goserelin; HR=0.659 (95% CI: 0.509-0.851)2
Time to chemotherapy was evaluated in a post hoc exploratory analysis and was defined as the time from randomization to the beginning of the first chemotherapy after discontinuing study treatment3
There was no prespecified statistical procedure controlling for type 1 error
MONALEESA-7 was a randomized, double-blind, placebo-controlled, phase III study of KISQALI + ET (NSAI or tamoxifen) + goserelin (n=335) vs placebo + ET (NSAI or tamoxifen) + goserelin (n=337) (ITT) in premenopausal patients with HR+/HER2- mBC who received no prior ET for advanced disease. KISQALI is not indicated for concomitant use with tamoxifen. Efficacy results are from a prespecified subgroup analysis of 495 patients who received KISQALI (n=248) or placebo (n=247) with an NSAI + goserelin and were not powered to show statistical significance. OS was a secondary end point; PFS was the primary end point.4,5
ET, endocrine therapy; HER2-, human epidermal growth factor receptor 2-negative; HR+, hormone receptor-positive; mBC, metastatic breast cancer; mTTC, median time to chemotherapy; PFS, progression-free survival.