MONALEESA-3 safety profile
KISQALI + fulvestrant in 1L/2L postmenopausal patients
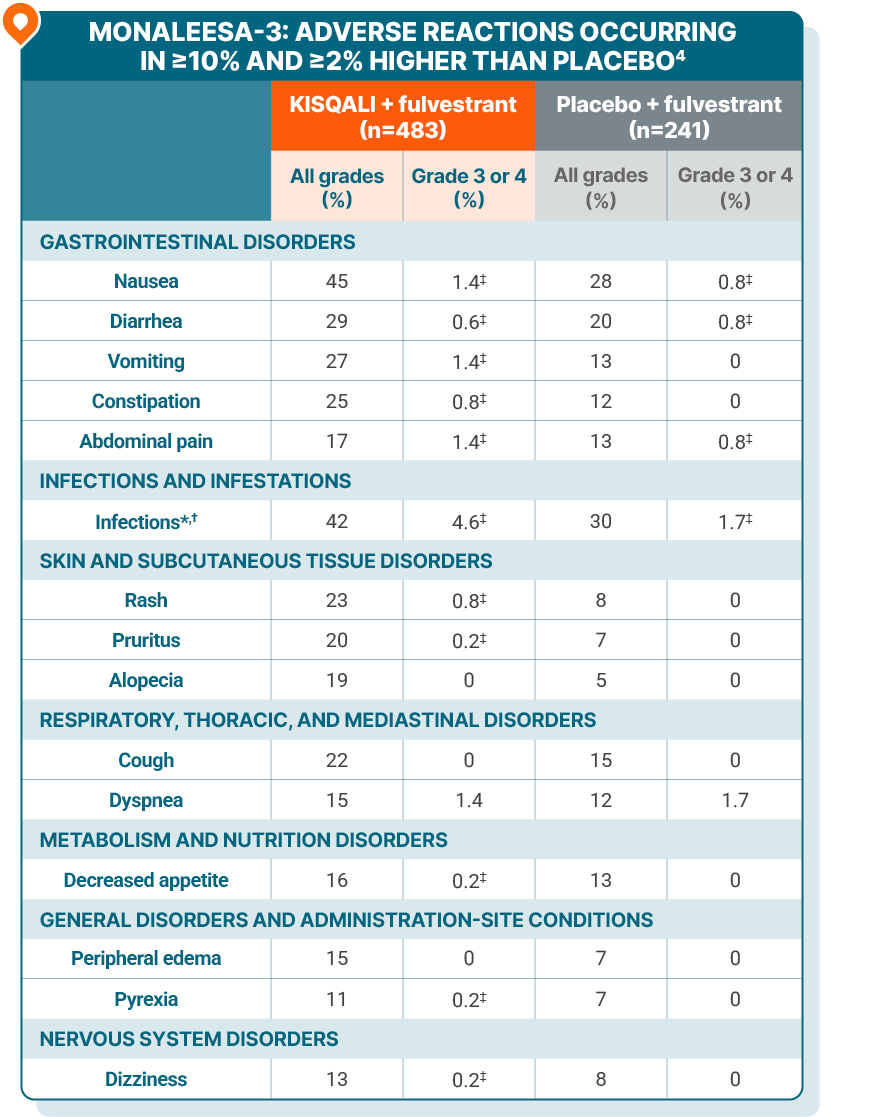
ADVERSE REACTIONS
Majority of adverse reactions were manageable and reversible1-3
Dose reductions due to ARs: 32% with KISQALI + fulvestrant4
Permanent discontinuations: 8% with KISQALI + fulvestrant4
Patients may require dose interruption, reduction, or discontinuation for ARs. Monitoring should include pulmonary symptoms, ECGs, serum electrolytes, LFTs, and CBCs. See Warnings and Precautions for risk of ILD/pneumonitis, SCARs, QT prolongation, hepatotoxicity, neutropenia, and embryo-fetal toxicity4
The most common ARs (≥20% on the KISQALI arm and ≥2% higher than placebo), including laboratory abnormalities, were decrease in leukocytes, decrease in neutrophils, decrease in lymphocytes, increase in creatinine, decrease in hemoglobin, increase in gamma-glutamyl transferase, increase in AST, nausea, increase in ALT, infections, decrease in platelets, diarrhea, vomiting, constipation, decrease in glucose serum, cough, rash, and pruritus4
Fatal ARs occurred in 1.2% of patients who received KISQALI. Fatal ARs in ≥0.1% of patients receiving KISQALI included cardiac failure, ventricular arrhythmia, pneumonia, acute respiratory distress, pulmonary embolism, and hemorrhagic shock (0.2% each)4
ARs in patients with visceral metastases receiving KISQALI were consistent with ARs in those without visceral metastases5
Grading according to CTCAE version 4.03.
*Infections included urinary and respiratory tract infections, gastroenteritis, and sepsis (1%).
†Includes the following fatal adverse reactions: pneumonia (n=1).
‡Only includes grade 3 ARs.
1L, first line; 2L, second line; ALT, alanine aminotransferase; AR, adverse reaction; AST, aspartate aminotransferase; CBC, complete blood count; CTCAE, Common Terminology Criteria for Adverse Events; ECG, electrocardiogram; ILD, interstitial lung disease; LFT, liver function test; SCAR, severe cutaneous adverse reaction.
LAB ABNORMALITIES
Lab abnormalities in MONALEESA-3
Scheduled blood tests and 2 upfront ECGs help to ensure your patients start KISQALI with confidence
Review the assessments schedule and the incidence of QT prolongation across clinical trials
The majority of adverse reactions with KISQALI were manageable and reversible
Review dose adjustment guidance